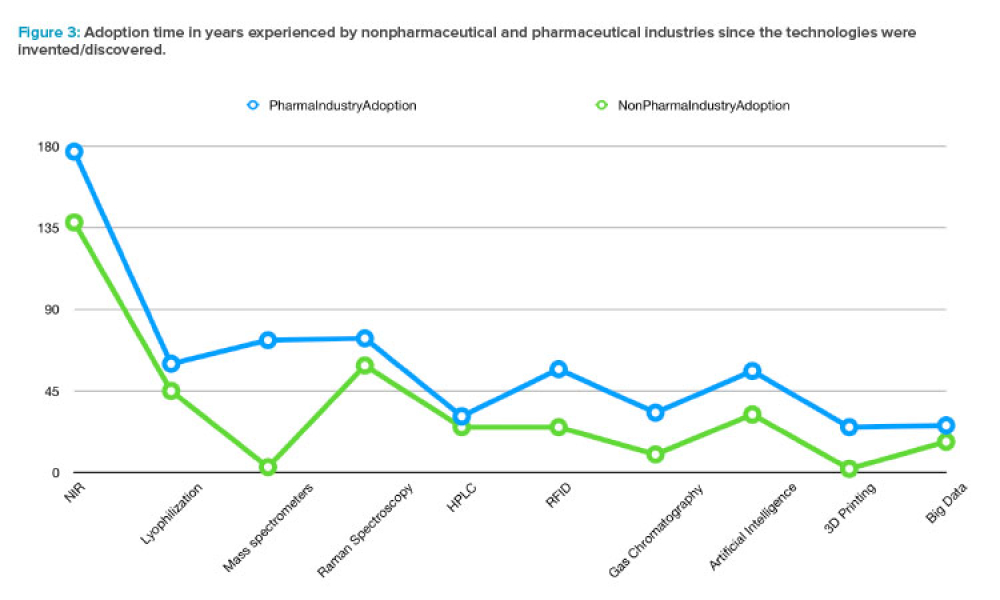
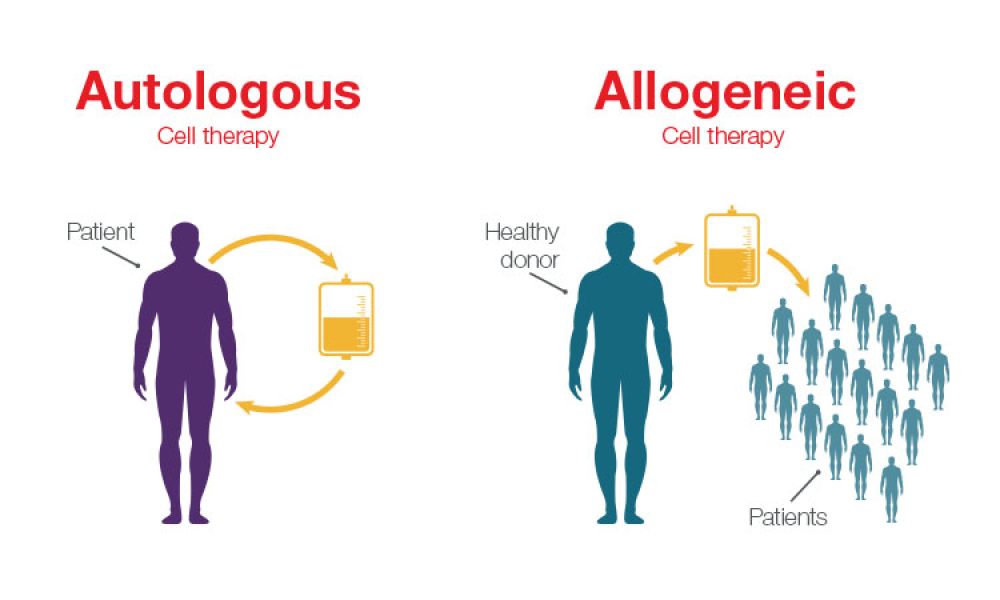
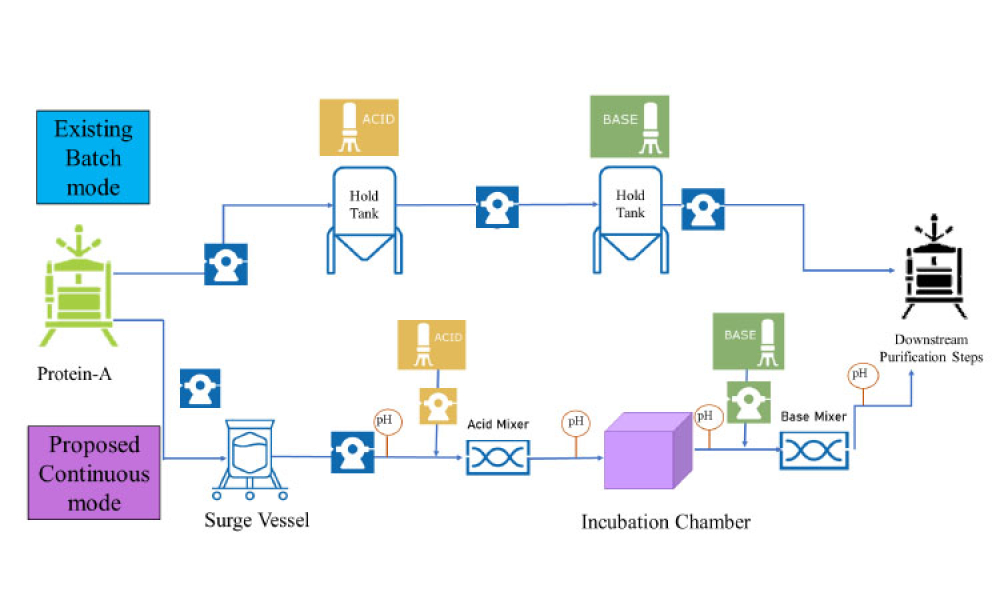
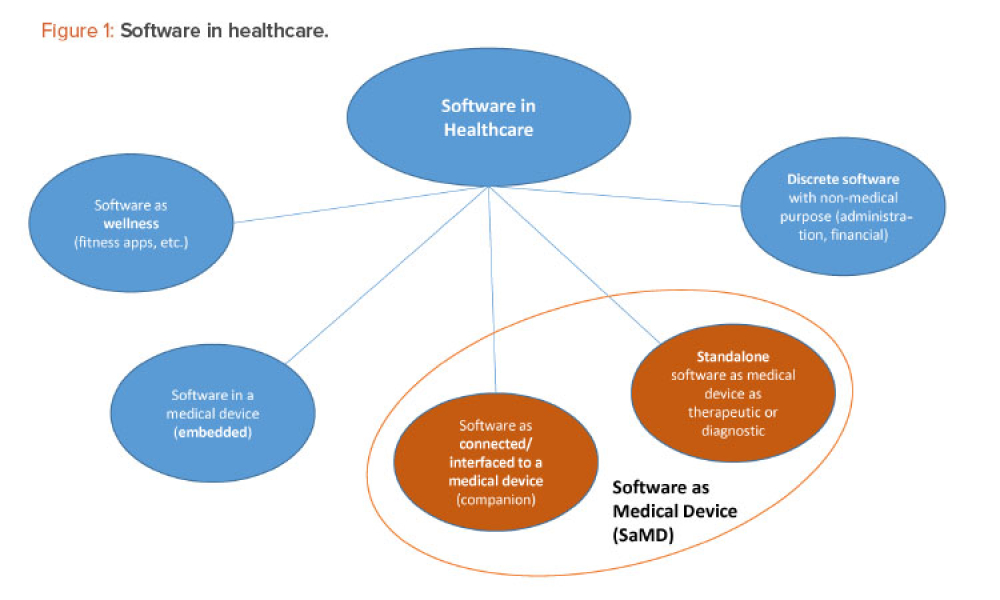
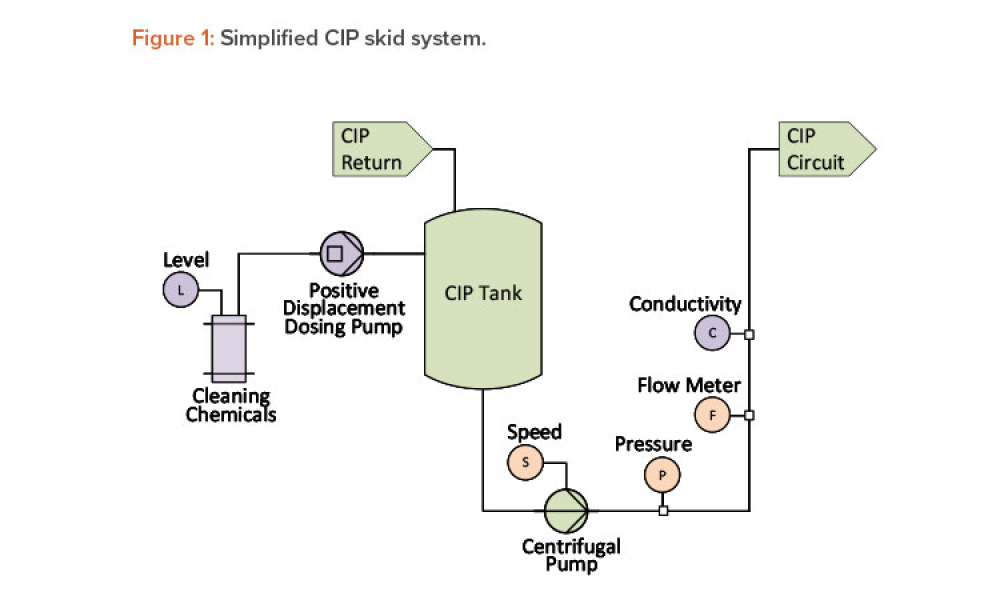
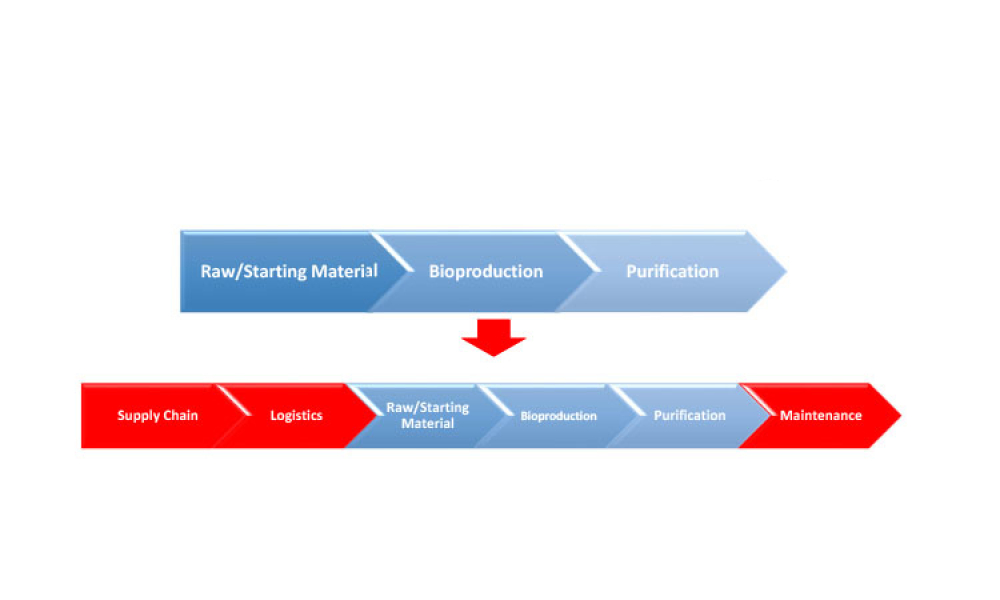
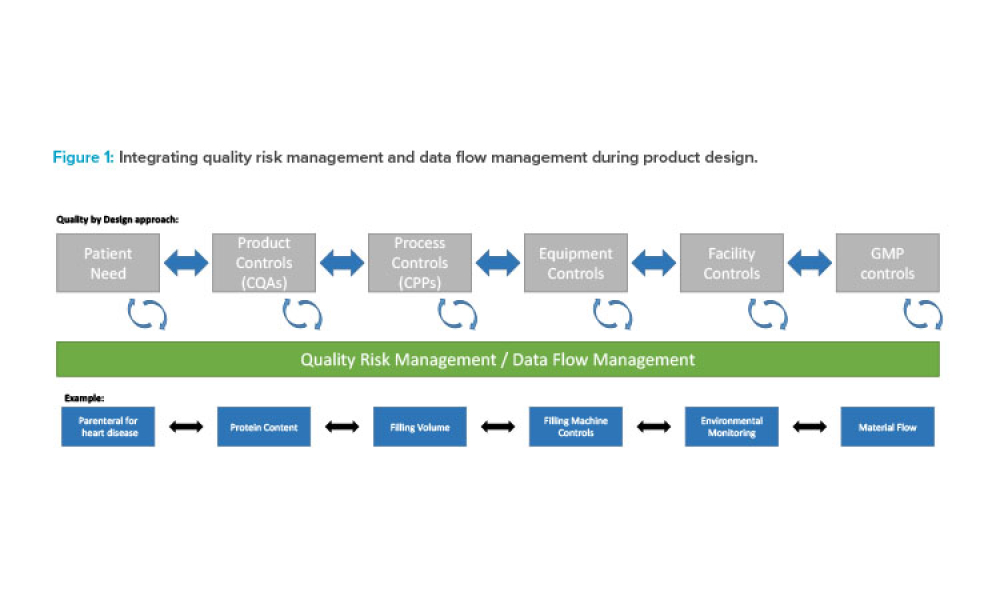
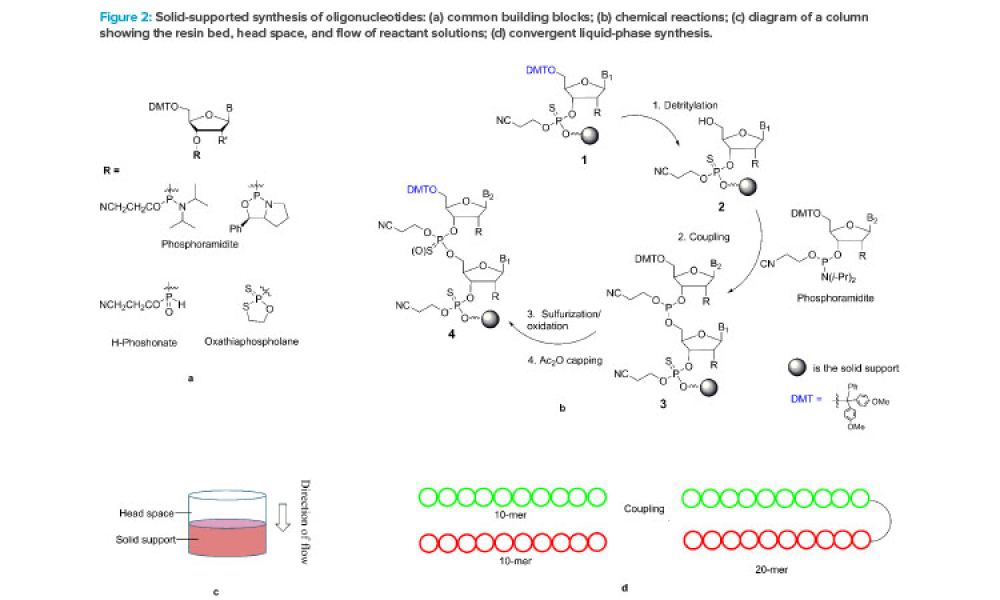
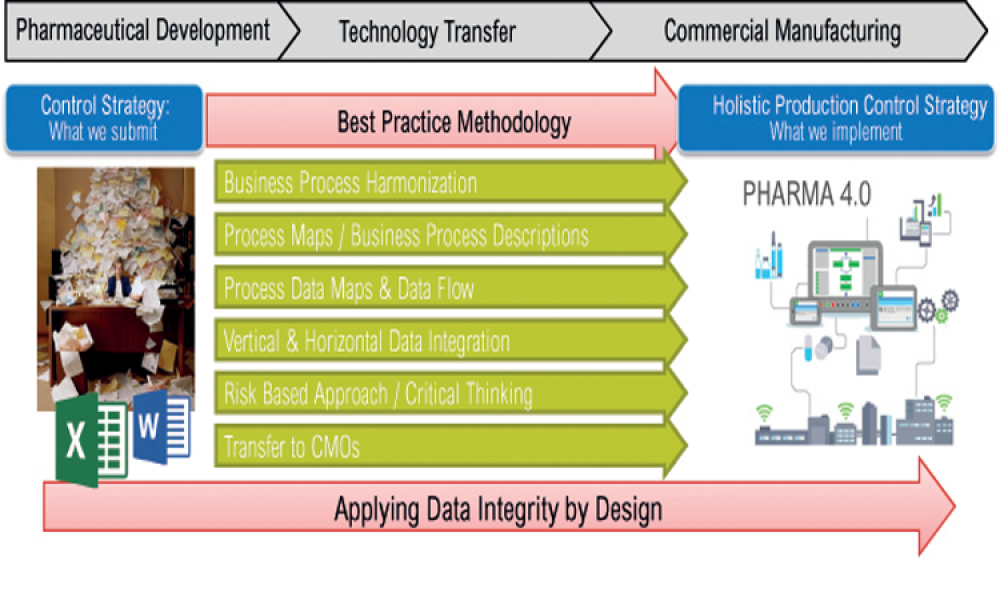
The ISPE Guide: Advanced Therapy Medicinal Products – Allogeneic Cell Therapy and the ISPE Guide: Advanced Therapy Medicinal Products – Recombinant AAV Comparability and Lifecyle Management were published earlier this year. These guides add to ISPE’s library of knowledge in the growing and ever-evolving advanced therapy medicinal products (ATMPs) field.
Transitioning from lab manufacture to commercialization presents significant challenges, no matter what area you’re working in. Cell and gene therapies are no different and production quantities must scale at multiple stages, first to support clinical trials and then when they reach the market. With uniquely challenging conditions required to protect medicinal integrity and exceptionally high...